ADH changes the way the kidneys react to blood flowing through them.The brain releases a hormone called antidiuretic hormone (ADH) (also known as arginine vasopressin (AVP)).In normal, healthy people the osmolality of the body's fluids is very closely regulated by the body. How does the body maintain fluid homeostasis? So fluid homeostasis means keeping the fluid the same all the time.

The word homeostasis comes from 'homeo' meaning alike or similar and 'stasis' meaning to remain the same. Mannitol is used for this effect to reduce brain swelling if you have had a head injury or brain surgery.įluid homeostasis is the term for the way the body keeps the osmolality of the body fluids within a very narrow range, all the time. They may also want to measure it to monitor the effects of medication designed to change the osmolality of your body tissues. It may even be due to you having overloaded your system by drinking too much water. This might be due to an illness or as a side-effect of medication. Osmolality is measured when doctors are concerned that your fluid balance may have gone awry for some reason. Occasionally the osmolality of stools (faeces) may also be tested. This is important as the cells won't work properly if this doesn't happen. This keeps the osmolality the same on both sides of the cell walls throughout the body. Permeable means it allows these particles and water to move freely in and out through the cell walls. This is because the walls of our cells are permeable to the microscopic particles (ions and anions) and to water. The serum osmolality is extremely close to the osmolality inside the cells that our body is made of.
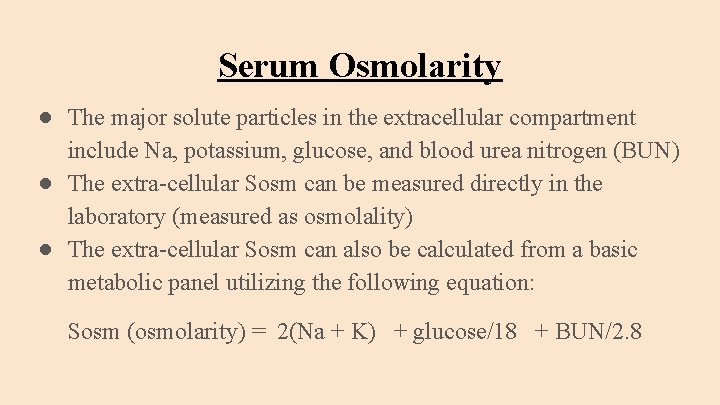
Often you will be asked for a sample of urine for urine osmolality testing too. The normal serum osmolality is 280-295 mOsm/kg. The particles that make up the osmolality of serum are glucose, sodium and potassium salts (Na + and K +) and urea. Serum is what is left from blood once the cells and proteins have been removed. The body fluid that we usually use to measure osmolality in humans is serum. What is the normal osmolality of body fluids? This is because the same weight of water takes up slightly more room as it warms up - it expands.īecause osmolarity changes with temperature, the term osmolality is preferred in medicine.

To take the same example as above, if you dissolve 100 g of salt in 1 kg of water, the osmolarity will decrease very slightly as the liquid warms up. Osmolarity will change depending on the temperature of the liquid. The measurement is given in millimoles per litre, or mmol/L for short. The number of particles is measured in millimoles, which is another measurement widely used in chemistry. The osmolarity of a fluid is a measure of the number of particles per litre of the liquid that they are dissolved in (the solute). This is the difference between osmolality and osmolarity. To take an example, if you dissolve 100 g of salt in 1 kg of water, the osmolality would be the same whether or not the water was near freezing point or at body heat. Osmolality does not depend on the temperature of the liquid. The measurement is given in milliosmoles per kilogram, or mOsmol/kg for short. The number of particles is measured in milliosmoles, which is a measurement widely used in chemistry. The osmolality of a fluid is a measure of the number of particles per kilogram of the liquid that they are dissolved in (the solute).
